The Food and Drug Administration ratified the first-ever cure that genetically changes a patient’s own cells to fight cancer, a distinguished sign that is expected to transform cure in the future years. The new treatment converts a patient’s cells into a “living drug” and attack the disease and trains them to identify. It is part of the quickly growing ground of immunotherapy that amplifies the immune system through medicines and other cures and has led to possibly even treats and long remissions.
The therapy sold as Kymriah and made by Novartis, was ratified for young people and children attacking type of leukemia (B-cell acute lymphoblastic leukemia) that has opposed standard cure. The FDA called the disease “deadly and destroying” and said the new cure loads an “unmet requirement.”
Novartis and other countries have been competing to expand gene therapies for other type of cancer and specialists envisage more ratifications in the near future. Dr. Scott Gottlieb, commissioner of the F.D.A. said that more than 550 types of experimental gene therapy were being studied.
We have a problems in this experiment. Because kymriah can have life–threatening side factors containing dangerous blobs in blood pressure, the FDA is needing that doctors and hospitals be specially educated and confirmed to manage it that stock a certain medicine required to quench violent reactions.
Kymriah, that will be given to patients just once and must be made exclusively for each will cost $475,000. Novartis added that if a patients doesn’t give a cure, there will be free. The company also give a financial help to people that have the disease and don’t able to pay it.
Considering the high cost during a telephone news speech, a Novartis official showed that bone marrow transplants that can treat some cases of leukemia price even more from $540,000 to $800,000.
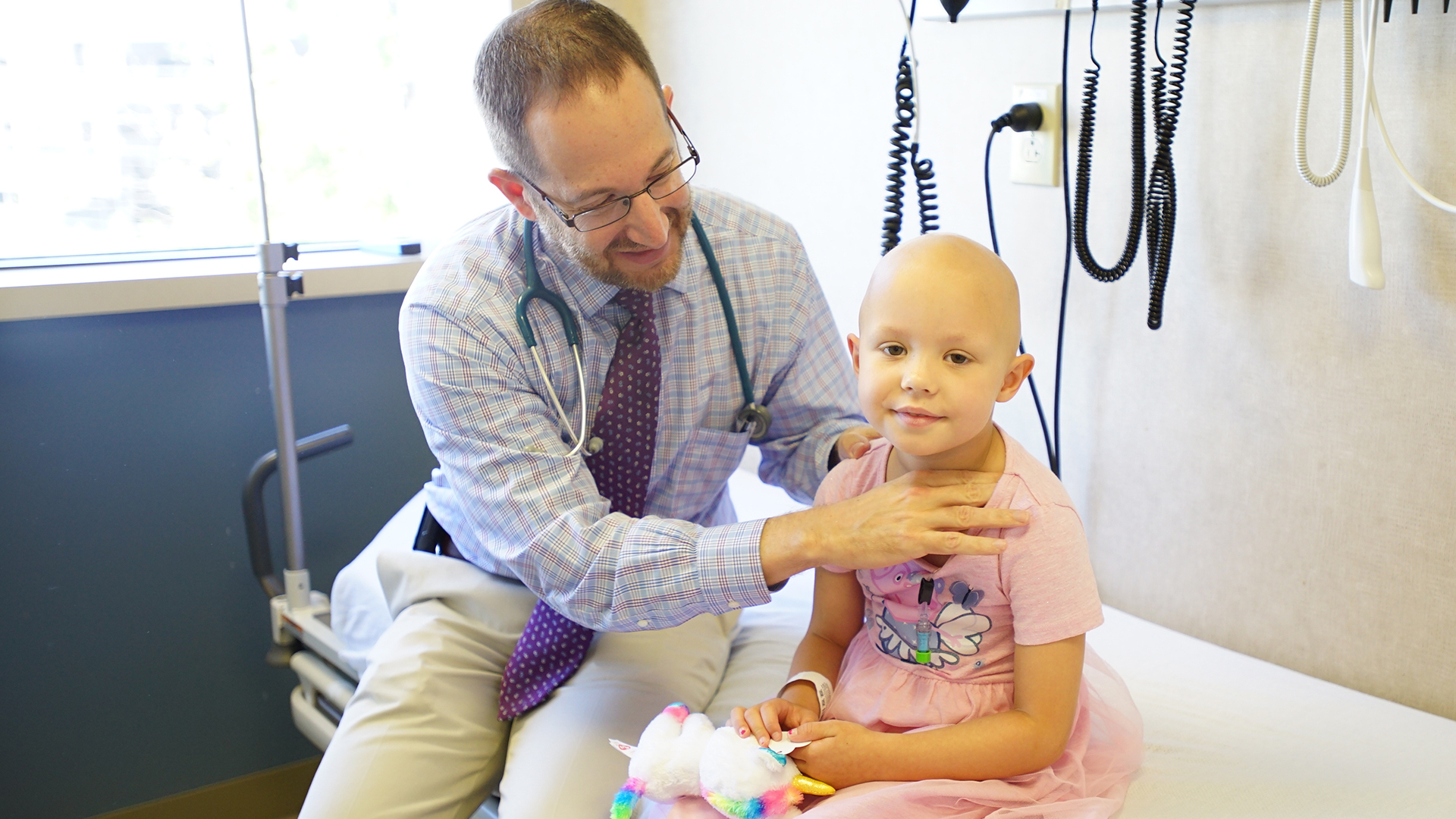
About 600 young adults and children a year in the United States would be candidates for the new cure.
The ratification was based on an experiment in 63 strongly ill young adults and children that had a remission level of 83 percent in three months, a high rate, given that recurrence or cure-resistant disease is often rapidly mortal. The cure was expanded by researchers at the University of Pennsylvania and licensed to Novartis. It was recognized in previous reports as CAR-T cell therapy, CTL019 or tisagenlecleucel.
The first child to take the cure was Emily Whitehead, who was 6 and near death from leukemia in 2012 when she was cured at the Children’s Hospital of Philadelphia. Now she is 12, she has been free of leukemia for more than 5 years.
To habituate Kymriah for individual patients, white blood cells that we called T cells will be omitted a patient’s bloodstream at an approved medical center, frozen, shipped to Novartis in Morris Plains, N.J., for genetic engineering and multiplying, frozen again and shipped back to the medical center to be abstracted into the patient. That processing is envisaged to take 22 days. Novartis said:
The cure would be available at primary network of 20 approved medical centers to be confirmed within a month, a number that would be expanded to 32 by the end of the year. Five centers will be ready to start extracting T cells from patients in three to five days.
Witness is being needed because the revved up T cell can feel an intense reaction, some time we called cytokine storm that can cause: lung congestion, low blood pressure, high fever, neurological problems and other life-threatening complications. Medical personal members require teaching to manage these reactions and hospitals told that before giving Kymriah to patients, they must be sure that they have the medicine required to cure the problems, tocilizumab called Actemra.
Dr. Kevin J. Curran, a pediatric oncologist at Memorial Sloan Kettering Cancer Center in Manhattan, said:
Our hospital was %99 of the path through the witness process and would soon be showing Kymriah. This is a big sample change, using this living medicine. It will give to you a lot of hope. This is the start.
He expected the treatment would work for other disease and cancer in future, not just for leukemia. The FDA’s ratification of Kymriah ushers in a new approach to the cure of cancer and other serious diseases. The company said in statements minding that the new therapy is the first gene therapy available in United States. Dr. Carl June, a chief in expanding the cure at the University of Pennsylvania, remembered that in 2010, when experiments illustrated that the first patients was leukemia-free a month after being treated and this is unbelievable for him and his colleagues.
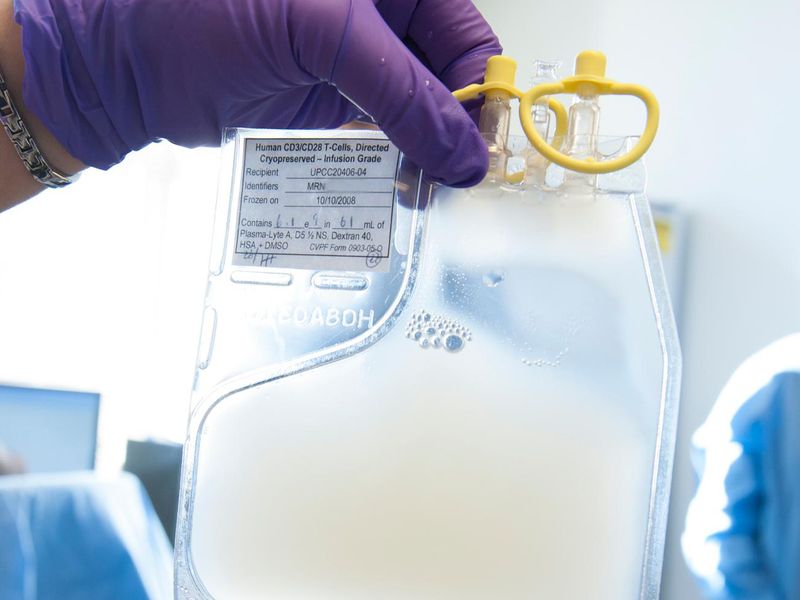
Dr. June said:
Now, I have to keep pinching myself to see that this happened, his voice breaking with emotion. It was so unlikely that this would ever be a commercially approved therapy and now it is the first gene therapy ratified in the United States. It’s so different from all the pharmaceutical patterns. I think the cancer world is forever changed.
Source link: https://www.nytimes.com/2017/08/30/health/gene-therapy-cancer.html
